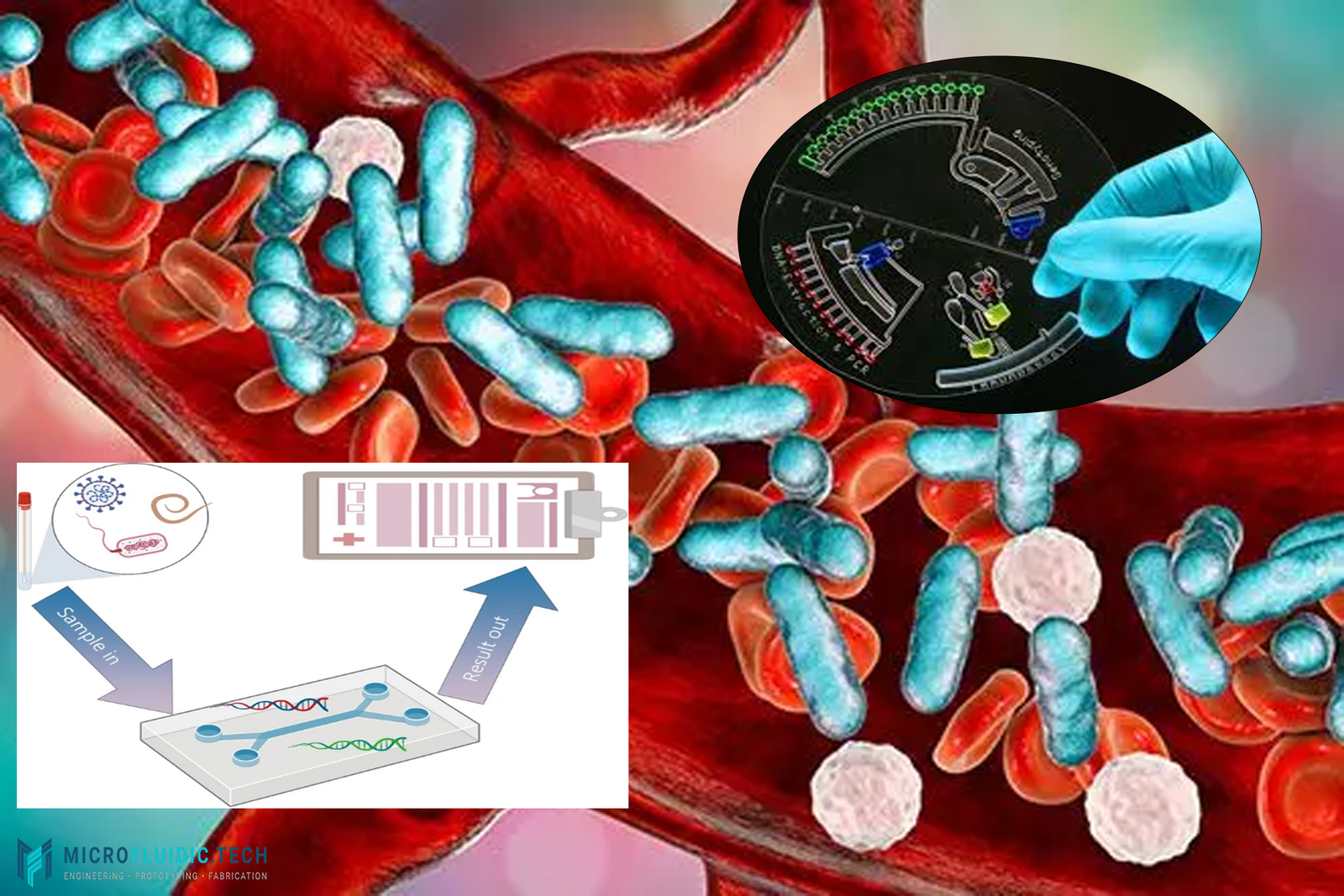
Sepsis is a life-threatening condition where early clinical deterioration often occurs before clear symptoms appear, making timely intervention difficult. A recent study published in Nature Communications introduces a machine learning–enabled centrifugal microfluidic platform that can predict sepsis-related clinical deterioration at the bedside, within hours of patient presentation.
This work highlights how microfluidics, molecular diagnostics, and artificial intelligence can converge to deliver truly actionable point-of-care solutions.
A Molecular Signature for Early Risk Prediction
The researchers identified a six-gene host-response signature, named Sepset, that reflects early immune cell reprogramming associated with sepsis progression. Unlike traditional biomarkers such as lactate or clinical scores like SOFA, this signature can predict worsening organ dysfunction within 24 hours, even before overt clinical decline.
The signature was developed using machine learning analysis of transcriptomic data from more than 3,000 patients and validated across multiple independent cohorts. It is pathogen-agnostic, performing consistently for both bacterial and viral sepsis, including COVID-19.
From the Lab to the Bedside: A Centrifugal Microfluidic Platform
To translate the biomarker into clinical practice, the team developed PREDICT, a stand-alone centrifugal microfluidic system that automates the entire workflow:
- Whole-blood RNA extraction
- Droplet digital PCR (ddPCR) amplification
- Fluorescence detection
- AI-based risk classification
Using only 50 µL of whole blood, the system delivers a risk assessment in under 3 hours, without requiring a centralized laboratory or specialized operators.
Clinical Performance
When tested on real patient samples, the microfluidic platform achieved:
- 92% sensitivity
- 89% specificity
- 88% overall accuracy
Notably, the molecular signature outperformed lactate measurements, and combining lactate with the gene signature did not improve prediction—underscoring the value of host-response molecular diagnostics.
Why This Matters for Microfluidics
This study represents a major step forward for translational microfluidics:
- Demonstrates true sample-to-answer automation
- Validates centrifugal microfluidics in real clinical settings
- Integrates AI, molecular biology, and microdevice engineering
- Enables decentralized, point-of-care decision making
Unlike many lab-on-a-chip demonstrations, this platform was validated using clinically annotated patient cohorts, setting a new benchmark for deployable microfluidic diagnostics.
Looking Ahead
Beyond sepsis, this microfluidic architecture can be adapted for immune profiling, infectious disease monitoring, and precision medicine applications. The study clearly shows that microfluidic platforms are evolving from analytical tools into predictive clinical systems.
Key Takeaway
By combining centrifugal microfluidics and machine learning, this work demonstrates how early, bedside prediction—not just diagnosis—can transform critical care. It is a compelling example of how microfluidic technologies are moving from the lab into real-world clinical impact.
https://www.nature.com/articles/s41467-025-59227-x