Introduction
Single-cell analysis — the ability to study individual cells instead of bulk populations — is one of the most transformative areas in biology and medicine. It enables high-resolution insights into cellular heterogeneity, disease mechanisms, developmental biology, precision medicine, and drug discovery. Many cutting-edge applications rely on isolating and manipulating one cell at a time. Microfluidics, with its ability to precisely control fluids at micrometer scales, has become a cornerstone technology for this purpose.
The Core Challenge
Traditional microfluidic cell-trapping systems typically use closed microchannels, where cells are guided through confined paths using pumps and pressure. These systems can trap single cells effectively, but the closed architecture makes it difficult or impossible to directly access the trapped cells for further analysis, sorting, or retrieval — a major limitation for workflows that require downstream processing like genetic sequencing or robotic picking.
What’s New: Open Microfluidic Single-Cell Trapping
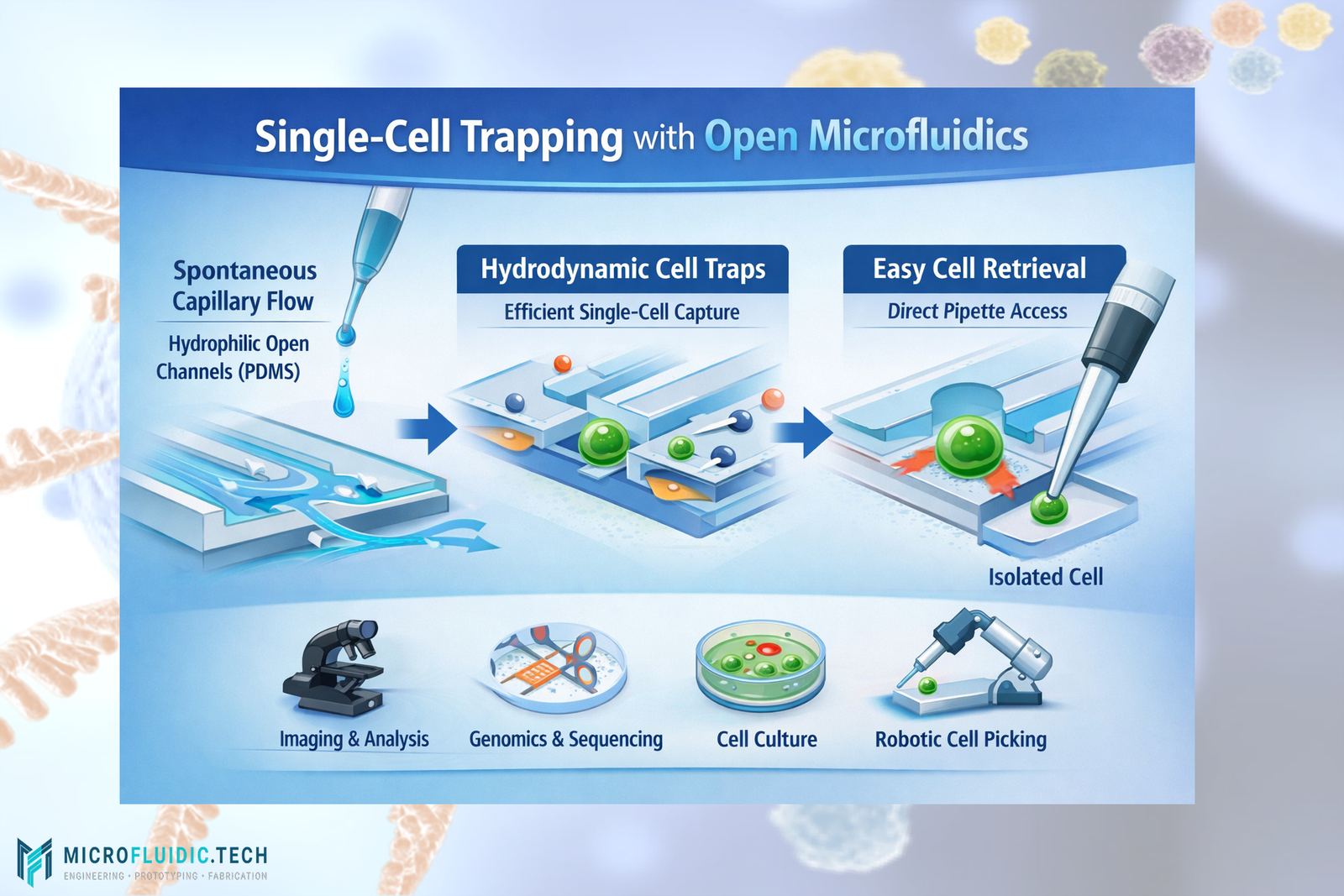
The study by Murakami and colleagues introduces an open microfluidics architecture that enables efficient single-cell trapping and direct access to the trapped cells from outside the channel — a significant advancement over traditional systems.
How the Microfluidic Device Works
Here’s how the technology is engineered:
-
Open Channel Design:
Instead of embedding cells within fully enclosed microchannels, the device features open microchannels etched into a PDMS (polydimethylsiloxane) substrate. These open channels allow fluid and cells to flow spontaneously without pumps and—critically—also allow open access to trapped cells for retrieval. -
Hydrophilic Surface Treatment:
Only the inside surfaces of the microfluidic channels are treated to become hydrophilic using specialized agents (e.g., Pluronic F-68 or polyvinyl alcohol), while the top surface remains untreated. This selective treatment ensures that spontaneous capillary flow (SCF)—the natural movement of liquid along hydrophilic surfaces—can reliably draw fluids and suspended cells into the microchannels. -
Hydrodynamic Traps:
The design incorporates deterministic hydrodynamic trapping structures inspired by classic microfluidic trap geometries. These microstructured regions create localized low-resistance pathways for cells to enter and become immobilized at specific sites within the open channels. -
Single-Cell Trapping & Efficiency:
When a drop of cell suspension is placed at the inlet, spontaneous capillary action draws the fluid through the narrow, cell-sized channels. The traps capture individual particles (e.g., microbeads) and cells with high efficiency — nearly 100% in many cases. -
Direct Cell Access & Retrieval:
Once cells are trapped, they remain exposed at the surface of the open channel. This unique feature allows researchers to use automated robotic systems, pipettes, or micromanipulators to retrieve individual cells directly without disassembling the device or flushing the system—something that closed channel systems cannot easily achieve.
Why This Matters in Microfluidics
✔️ Bridging Micro and Macro Worlds
This open microfluidic platform bridges the gap between precise microscale fluid control and macroscale biological workflows, such as robotic single-cell picking and high-throughput analyses common in biological labs.
✔️ Simplified Operation
With spontaneous capillary flow driving fluid movement, there’s no need for external pumps or complicated pressure setups, making the system more accessible for routine lab use and scalable screening applications.
✔️ Expanded Compatibility
Exposing the cells after trapping allows easy integration with:
-
Microscopy and live imaging
-
Fluorescent sorting
-
Single-cell sequencing pipelines
-
Cell culture and proliferation studies
All of these benefit from direct pipette or robotic access to isolated cells — a major advantage over traditional microfluidic traps.
✔️ Potential for High-Throughput
Because multiple hydrodynamic traps can be arrayed within the open microchannel network, this device promises increased throughput for single-cell experiments while retaining individual cell access.
Implications for Research & Industry
This open microfluidic single-cell trapping approach contributes to ongoing trends in the microfluidics field:
🔹 Single-cell genomics and transcriptomics
🔹 Cell line selection and screening in drug discovery
🔹 Precision cell therapy development
🔹 Stem cell research and rare cell isolation
By simplifying the trap and access mechanism, this method reduces barriers for labs without specialized microfluidic experience and supports hybrid workflows that combine microfluidic precision with traditional cell biology techniques.